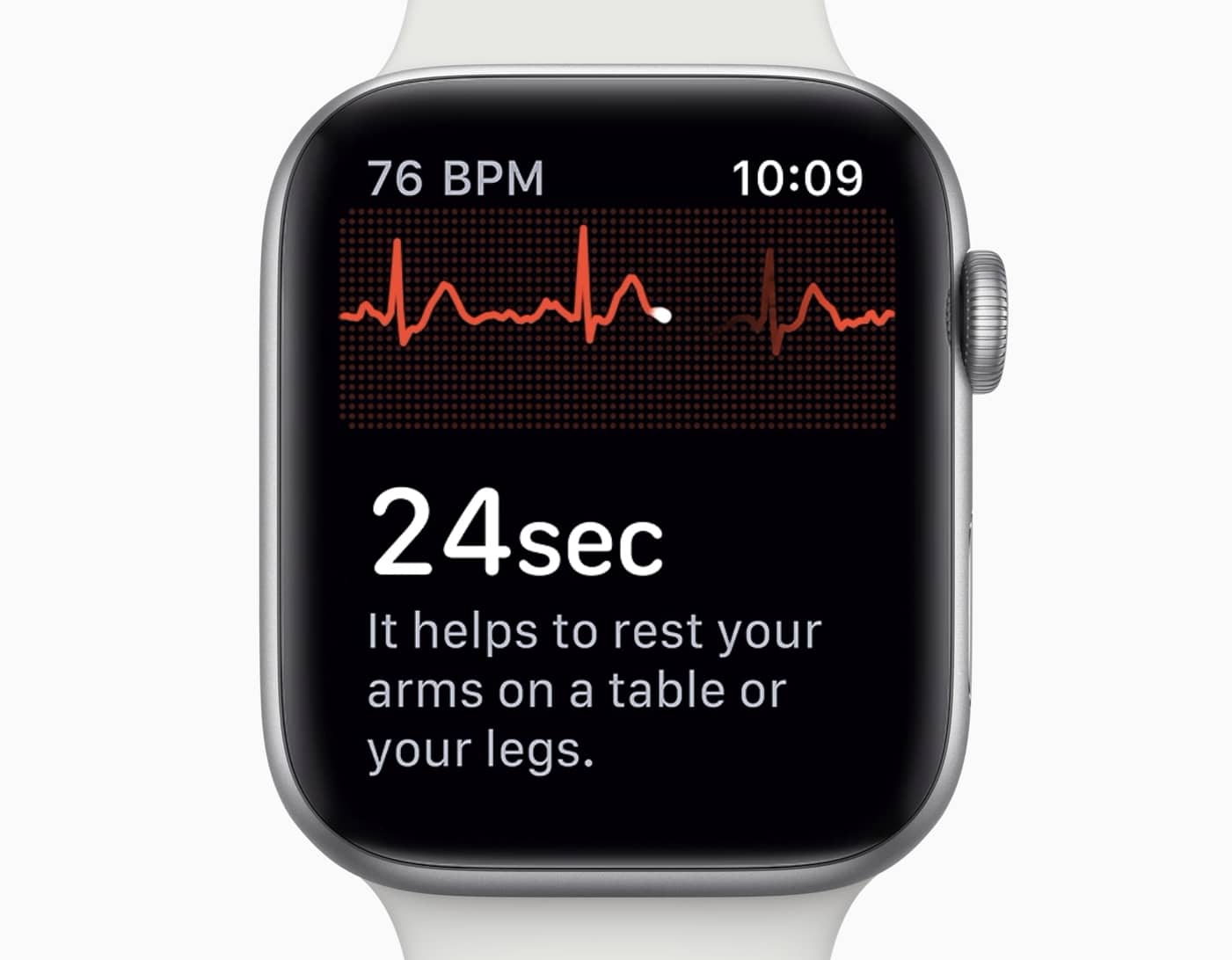
Apple Watch Series 4’s incredible ability to offer electrocardiograms (ECG) is one of the biggest reasons to upgrade this year. But you can’t use it yet.
Those in the U.S. will be able to get their hands on the feature before the end of 2018, but for those in other countries, the wait could be a lot longer. If you live in the U.K., you may even have to wait “years,” one report warns.
You can’t just sell a device with an ECG feature to consumers without the necessary clearance from regulatory bodies. That’s one of the reasons why it has taken years for Apple to implement this feature into the Watch.
In the U.S., it’s the Food and Drug Administration (FDA) that gives devices like this the go-ahead — and it has already cleared Apple Watch Series 4. In other countries, Apple must go through a similar approval process all over again.
It could take years to bring ECGs to the U.K.
In the U.K., Apple must obtain clearance from the Medicines & Healthcare Products Regulatory Agency (MHRA). 9to5Mac got in touch with them to find out how the approval process works for devices like the Apple Watch — and it doesn’t sound good.
The first step of the process would be for the MHRA to examine the information Apple supplies and ensure that Apple Watch lives up to its promises. If it is satisfied, the process could be fairly straightforward, and users in the U.K. could have access to ECGs relatively soon.
However, Apple could be forced to carry out a “clinical investigation” that evaluates the effectiveness of its ECGs — and the MHRA would probably not accept data from the trials Apple has already carried out.
Apple Watch may have to prove itself again
“You may need to carry out a clinical investigation as part of the process to obtain a CE marking for your medical device,” the MHRA explains. “You must inform MHRA if you are planning to do this at least 60 days before starting your investigation, [providing] some basic details about the investigational device, the intended population, the type of study, and estimated application date.”
Unless Apple already informed the MHRA of its trials 60 days in advance, then — and received approval long before they began — any data it has already collected is no good.
Assuming Apple would need to apply for a clinical investigation through the MHRA, it would have to wait 60 days for approval. Only after it has received that can the lengthiest step in the process begin.
“The last factor could be the most time-consuming and could potentially add years onto the CE marking process,” the MHRA explains.
Apple could be way ahead
This is the worst case scenario, of course.
It’s possible Apple has already started the process with the MHRA and is a lot closer to getting a stamp of approval. It’s also possible Apple has applied for approval from another regulatory body in the European Union, which would cover Apple Watch for the U.K., too (while the U.K. is still in the E.U.).
If none of this has taken place yet, however, those of us on the other side of the Atlantic may not be getting an ECG feature at all with Series 4.
We’ve contacted Apple for comment and we’ll update this post if we get a response.