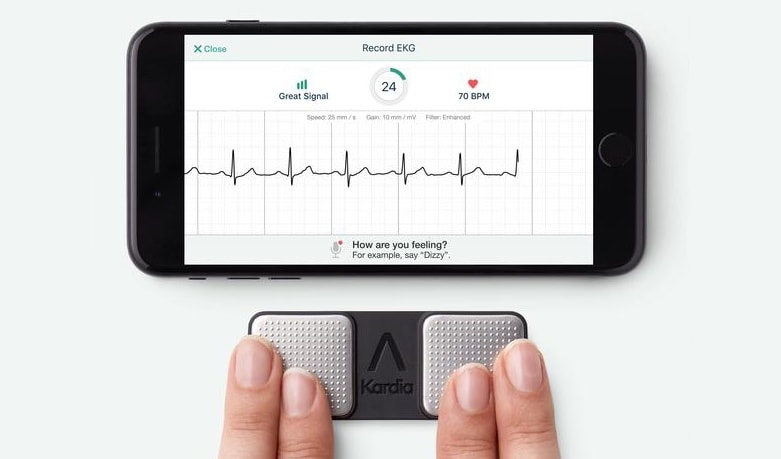
AliveCor‘s KardiaMobile has been cleared by the FDA to detect the three most common heart arrhythmias. It’s the only personal ECG cleared to do so.
That puts this pocket-size phone accessory well ahead of the Apple Watch, which can only detect one type of heart problem.
“Until today, patients have been frustrated when devices label their ECG reading as ‘unclassified’ or ‘inconclusive,’” said AliveCor CEO, Ira Bahr. “Starting today, KardiaMobile is the first personal ECG device that can begin to materially reduce the number of those determinations.”
KardiaMobile: Testing 1, 2, 3….
This accessory could already detect Atrial Fibrillation (AFib), and notify the user if they were experiencing Normal Sinus Rhythm. The US Food and Drug Administration has just cleared AliveCor’s heart sensor for Bradycardia and Tachycardia, which literally mean “slow heart” and “fast heart”, respectively.
While the latter two are often benign, they can be indicative of heart disease or other health conditions, such as thyroid disease. AFib, on the other hand, always a problem. It often goes undetected and brings increased risk of heart failure and stroke.
The Apple Watch Series 4 is only cleared by the FDA to detect AFib.
KardiaMobile available now
KardiaMobile is $99 from the AliveCor website. It’s compatible with every current Apple handset, and older models going back to the iPhone 5. It can also be used with an iPad, as well as many Android devices.
While not as portable as an Apple Watch, this item is 3.2 inches by 1.25 in. by .14 in., and 0.6 ounces. It comes with a clip to attach this product to the back of a phone.